CBSE Class 12 Atoms Multiple Choice Questions with Answers. MCQ Questions Class 12 Atoms with Answers Is Prepared Based on Latest Exam Pattern. Students can solve NCERT Class 12 Atoms MCQs with Answers to know their preparation level.
Students who are searching for NCERT MCQ Questions for Class 12 Atoms with Answers are compiled here to get good practice on all fundamentals. Know your preparation level on MCQ Questions for Class 12 Atoms with Answers. You can also verify your answers from our provided MCQ Class 12 Atoms with Answers. So, ace up your preparation with MCQ of Class 12 Physics Examinations.
MCQ Questions Class 12 Atoms with Answers - Set - 2
Question 1:
Energy levels A, B, C of a certain atom correspond to increasing value of energy i.e., EA > EB > EC. If λ1, λ2 and λ3 are the wavelengths of radiation corresponding to transition C to B, B to A and C to A respectively, which of these of the following is correct?
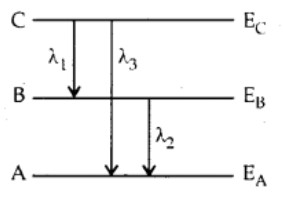
(a) λ3 = λ1 +d λ2
(b) λ3 = (λ1λ2)/(λ1+λ2)
(c) λ1 + λ2 + λ3 = 0
(d) λ23 = λ21 and λ22
Correct Answer – (B)
Question 2 :
If the electron in hydrogen atoms is excited to n = 5 state, the number of different frequencies of radiation which may be emitted is:
(a) 4
(b) 10
(c) 8
(d) 5
Correct Answer – (B)
Question 3 :
For an electron orbit to be non-radiating, it should be
(a) such that the angular momentum should be integral multiple of h.
(b) circular in nature
(c) elliptical in nature
(d) none of these
Correct Answer – (A)
Question 4 :
How does the energy difference between two consecutive energy levels vary on the quantum number n increases?
(a) does not change
(b) decrases
(c) increases
(d) may increase or decrease.
Correct Answer – (B)
Question 5 :
The ratio of the energy of the electron in first orbit to that in the second orbit is
(a) ¼
(b) ½R
(c) 2
(d) 4
Correct Answer – (D)
MCQ Questions Class 12 Atoms with Answers
Question 6 :
(a) ½
(b) ¼
(c) 4
(d) 2
Correct Answer – (A)
Question 7 :
Which of the following type ot radiation is not emitted by the electronic structure of atoms :
(a) X-rays
(b) Visible light
(c) γ-rays
(d) Ultraviolet light.
Correct Answer – (C)
Question 8 :
According to classical theory, Rutherford atom is
(a) stable
(b) unstable
(c) metastable
(d) semistable
Correct Answer – (B)
Question 9 :
When an electron jumps from some outer orb it to the innermost orbit in the hydrogen atom, the spectral line belongs to
(a) Lyman series
(b) Balmer series
(c) Paschen series
(d) Pfund series
Correct Answer – (A)
Question 10 :
To explain fine structure of spectrum of hydrogen atom, we must consider.
(a) a finite size of nucleus.
(b) the presence of neutrons in the nucleus.
(c) spin angular momentum.
(d) orbital angular momentum.
Correct Answer – (B)
- NCERT Solutions Class 11 Chemistry Chapter 1 : Some Basic Concepts of Chemistry
- NCERT Solutions Class 11 Chemistry Chapter 2 : Structure Of The Atom
- NCERT Solutions Class 11 Chemistry Chapter 3 : Classification of Elements and Periodicity in Properties
- NCERT Solutions Class 11 Chemistry Chapter 4 : Chemical Bonding and Molecular Structure
- NCERT Solutions Class 11 Chemistry Chapter 5 : States of Matter
- NCERT Solutions Class 11 Chemistry Chapter 6 : Thermodynamics
- NCERT Solutions Class 11 Chemistry Chapter 7 : Equilibrium
- NCERT Solutions Class 11 Chemistry Chapter 8 : Redox Reactions
- NCERT Solutions Class 11 Chemistry Chapter 9 : Hydrogen
- NCERT Solutions Class 11 Chemistry Chapter 10 : The s-Block Elements
- NCERT Solutions Class 11 Chemistry Chapter 11 : The p-Block Elements
- NCERT Solutions Class 11 Chemistry Chapter 12 : Organic Chemistry: Some Basic Principles and Techniques
- NCERT Solutions Class 11 Chemistry Chapter 13 : Hydrocarbons
- NCERT Solutions Class 11 Chemistry Chapter 14 : Environmental Chemistry